Copper and copper alloys are one of the major groups of commercial metals. They offer a wide range of properties, including excellent electrical and thermal conductivity, outstanding corrosion resistance, good strength and fatigue resistance, and appearance. They can be readily worked, brazed and welded.
- Electrical conductivity: copper has the highest conductivity of the engineering metals. Silver or other elements may be added to increase strength, softening resistance or other properties without major loss of conductivity.
- Thermal conductivity: this property is similar to electrical conductivity. Alloys of copper may be used for this property, where good corrosion resistance compensates for loss of conductivity with increased alloying.
- Colour and appearance: many of the copper alloys have a distinctive colour, which may change as the object weathers. For most of alloys it is easy to prepare and maintain the surface to a high standard, even in adverse corrosion conditions. Many of the alloys are used in decorative applications, either in their native form or after metal plating. The alloys have specific colours, ranging from the salmon pink of copper through yellow, gold and green to dark bronze in the weathered condition. Atmospheric exposure can produce a green or bronze surface, and prepatinated alloys are available in some product forms.
- Ease of fabrication: most of the alloys can be easily cast, hot or cold formed, machined, joined etc. These alloys are often the standard against which other metals are compared.
Common Types of Metal Alloys
Copper
In its pure, unalloyed state is soft, provides high electrical and thermal conductivity and has excellent corrosion resistance. There are various grades of unalloyed copper, which differ in the amount of impurities they contain. Oxygen-free coppers are used specifically in applications requiring high conductivity and exceptional ductility.
Brasses
Brasses are alloys made from copper and zinc, they exhibit good strength and ductility and are easily cold worked, properties which improve with increased zinc content up to 35%. Brass coloration ranges from red to golden yellow, depending on the amount of zinc the alloy contains. Gilding Metal, Commercial Bronze, Jewelry Bronze, Red Brass and Cartridge Brass are common names given to brass alloys with specific zinc contents. Brasses containing between 32% and 39% zinc exhibit excellent hot working characteristics but limited cold workability. Brasses containing more than 39% zinc, such as Muntz Metal, have high strength and lower ductility at room temperature than alloys with less zinc. Brasses are known for their ease of fabrication by drawing, high cold-worked strength and corrosion resistance. Brasses are routinely blanked, coined, drawn and pierced to produce springs, fire extinguishers, jewelry, radiator cores, lamp fixtures, ammunition, flexible hose and the base for gold plate. Brasses have excellent castability. Cast brasses are used as plumbing fixtures, decorative hardware, architectural trim, low pressure valves, gears and bearings.
Tin Brasses
Tin Brasses are alloys made from copper, zinc (2% to 40%) and tin (0.2% to 3%). This family of alloys includes admiralty brasses, naval brasses and free-machining tin brasses. These alloys are used to make high-strength fasteners, electrical connectors, springs, corrosionresistant mechanical products, marine hardware, pump shafts, and corrosion-resistant screw machine parts. They provide increased corrosion resistance, lower sensitivity to dez- incification and higher strength compared with straight brasses. They possess good hot forgeability and good cold formability. These materials have moderate strength, high atmospheric and aqueous corrosion resistance and excellent electrical conductivity.
Silicon Bronzes
Silicon Bronzes are part of the subgroup of high-strength brasses. They contain less than 20% zinc and up to 6% silicon and are solid solution strengthened. Silicon red brasses are used for valve stems where corrosion resistance and high strength are critical. Included in this category are the silicon red bronzes, which are similar to silicon red brasses except for their very low concentrations of zinc. They are used to make bearings, gears and intricately shaped pump and valve components.
Nickel Silvers
Nickel Silvers, also called nickel brasses, are alloys containing copper, nickel, and zinc. Though they do not contain silver, they have an attractive silver luster, moderately high strength and good corrosion resistance. They are used to make food and beverage handling equipment, decorative hardware, electroplated tableware, optical and photographic equipment and musical instruments.
Copper Nickel
Copper Nickel alloys contain anywhere from 2% to 30% nickel, are highly corrosion-resistant and thermally stable. The addition of iron, chromium, niobium and/or manganese can improve their strength and corrosion resistance. They are virtually immune to stress corrosion cracking and exhibit high oxidation resistance in steam and moist air. The higher nickel alloys are well known for their corrosion resistance in sea water as well as resistance to marine biofouling. They are used to make electrical and electronic products, tubes for condensers in ships, on offshore platforms and in power plants, and various other marine products including valves, pumps, fittings and sheathing for ship hulls.
Phosphor Bronzes
Phosphor Bronzes or tin bronzes as they are sometimes called, contain between 0.5% and 11% tin and 0.01% to 0.35% phosphorous. Tin increases their corrosion resistance and tensile strength; phosphorous increases wear resistance and stiffness. Phosphor bronzes have superb spring qualities, high fatigue resistance, excellent formability and solderability, and high corrosion resistance. They are used primarily for electrical products; other uses include corrosion resistant bellows, diaphragms and spring washers.
Aluminum Bronzes
Aluminum Bronzes contain 6% to 12% aluminum, up to 6% iron and nickel and provide high strength and excellent corrosion and wear resistance. Solid solution strengthening, cold work and precipitation of an iron rich phase contribute to these characteristics. High aluminum containing alloys can be quenched and tempered. Aluminum bronzes are used in marine hardware, shafts and pump and valve components for handling seawater, sour mine waters, nonoxidizing acids, and industrial process fluids. They are also used as heavy duty sleeve bearings and machine tool ways. Aluminum bronze castings have exceptional corrosion resistance, high strength, toughness and wear resistance. They also exhibit good casting and welding characteristics.
Specialty Copper Alloys
Specialty Copper Alloys, based, for example, on the copper-nickel-silicon and copper-nickel-tin systems, provide unique combinations of properties due to their intrinsic precipitation hardening capability. Their high strength coupled with good formability, thermal stability and electrical conductivity make them appropriate for use in electrical and electronic connectors and hardware. These alloys have designations throughout the UNS system based upon their composition. As we have seen, copper and its alloys constitute a broad range of chemical compositions and are employed widely in applications that enable and enhance our everyday lives. Each application makes effective use of copper’s attributes: strength, conductivity, color, formability, joinability and thermal stability.
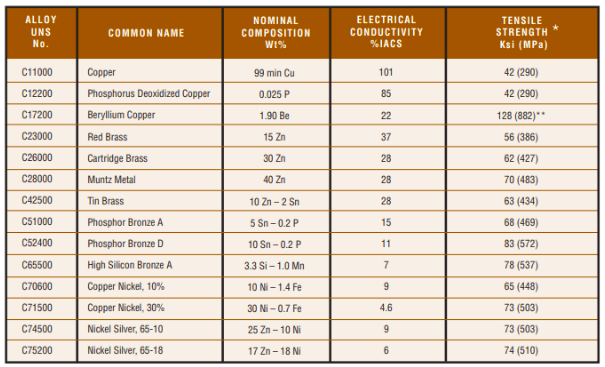
Compositions & Properties of the More Common Copper Alloys